Actionable Insights that Amplify Impact
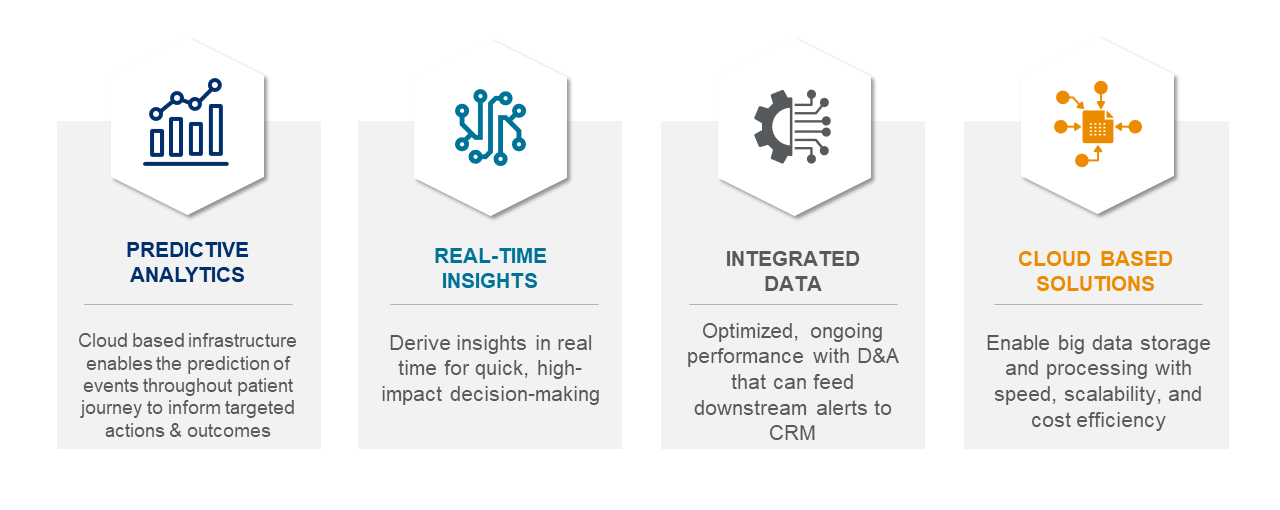
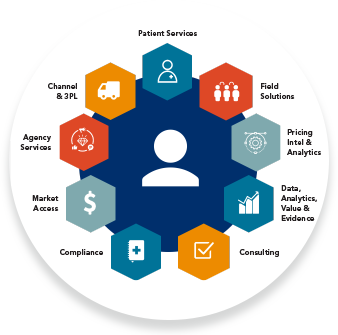
ACTICS by EVERSANA is the premier tech-enabled solution built to deliver end-to-end commercial success for life science companies. Leveraging a combination of proprietary artificial intelligence (AI) and machine learning (ML) technologies, infrastructure, data and subject matter expertise, ACTICS was developed to address critical challenges throughout the product life cycle and improve patient outcomes.